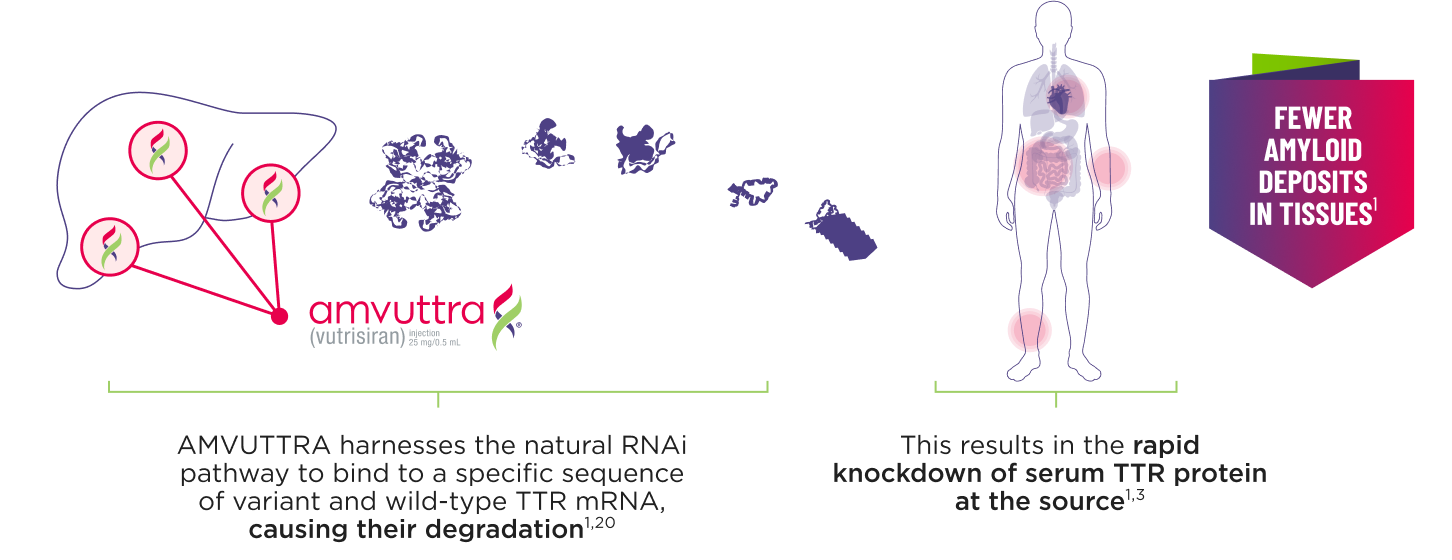
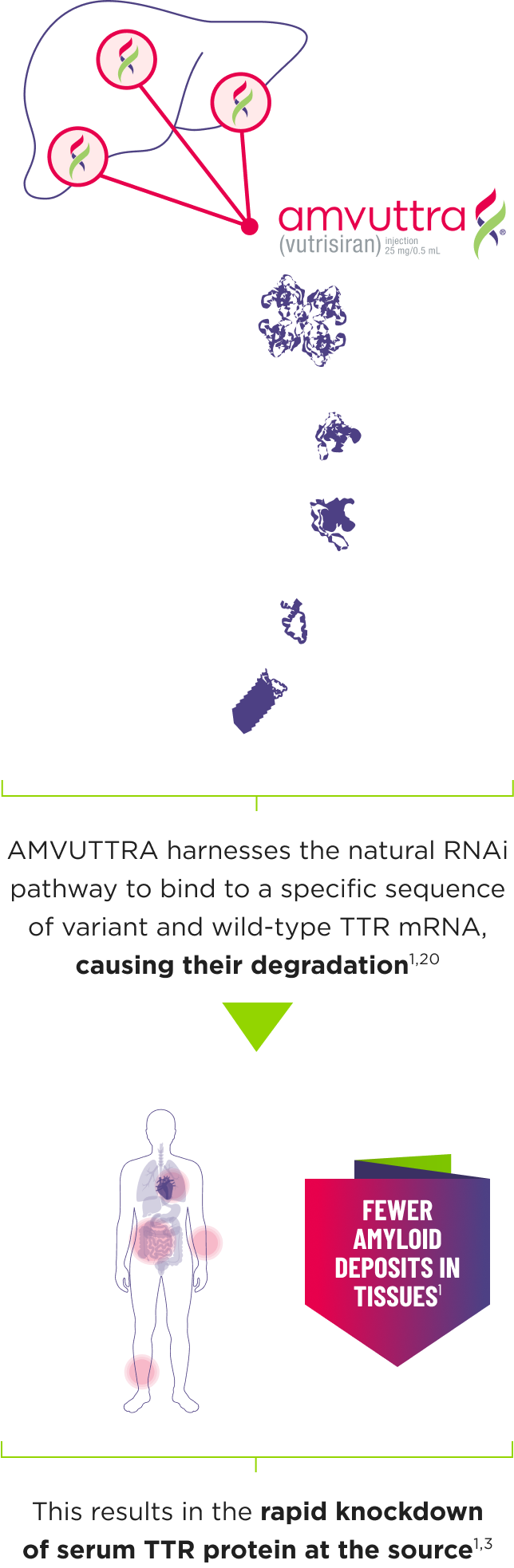
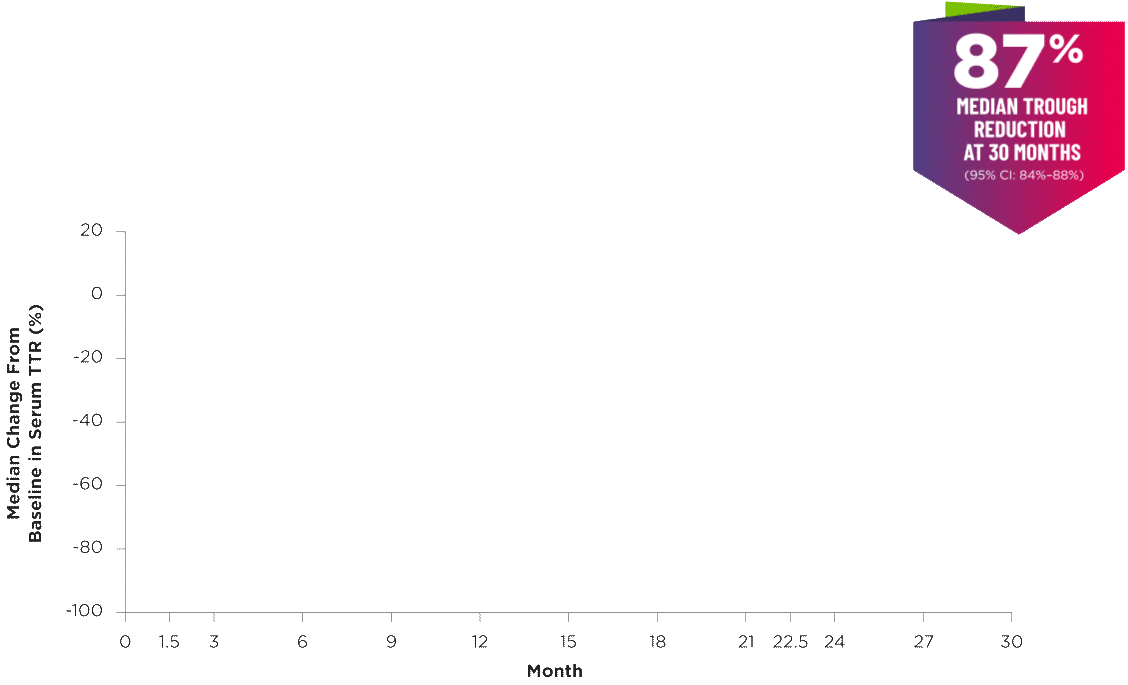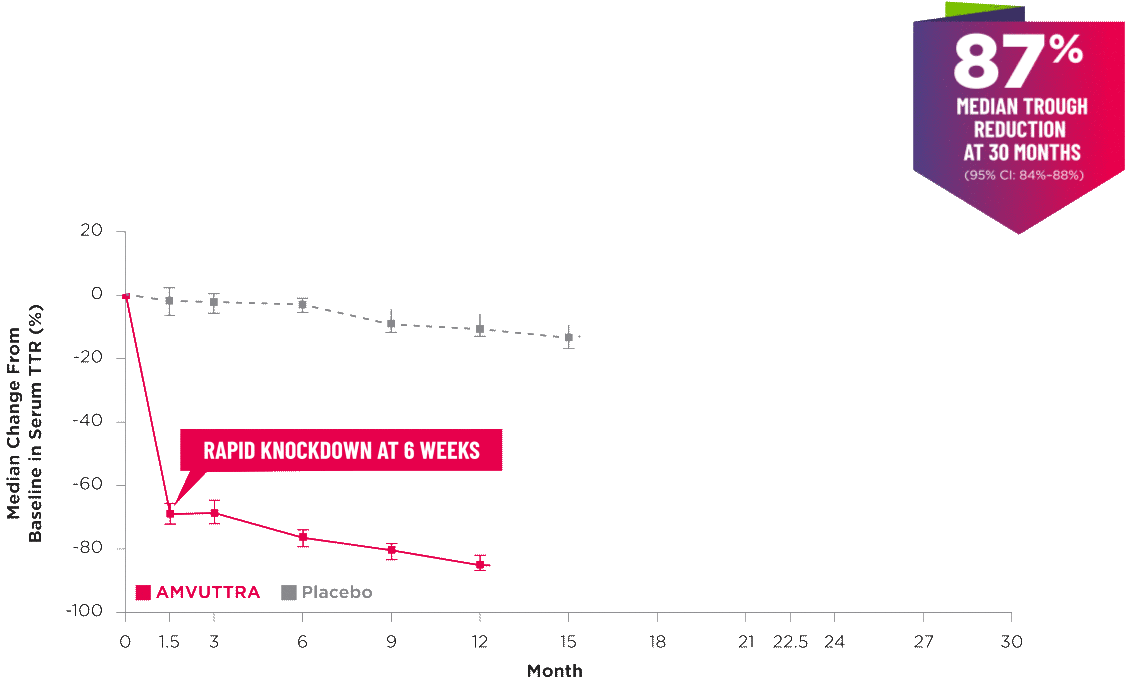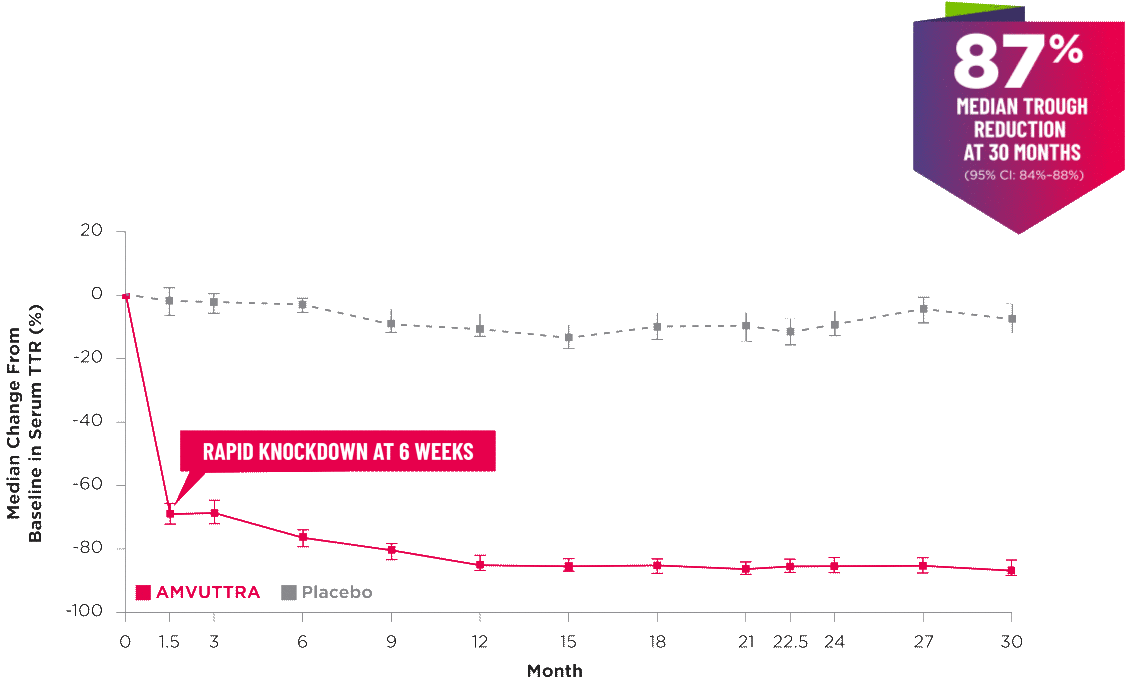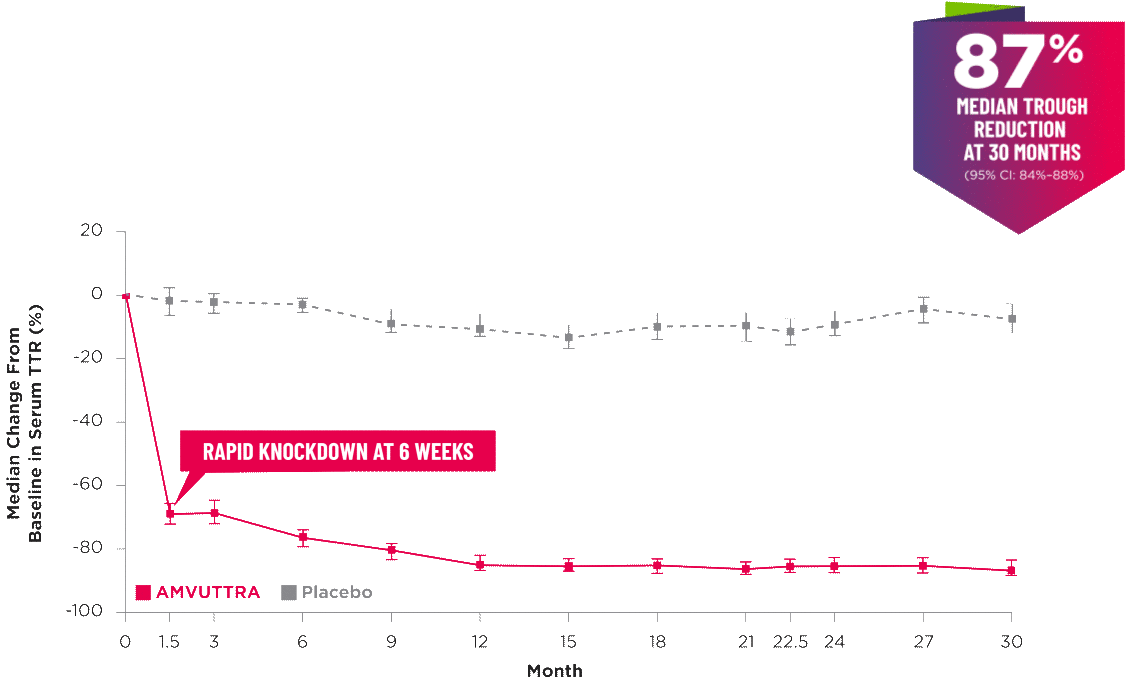How AMVUTTRA® Works
Suppress TTR production at the source with AMVUTTRA1
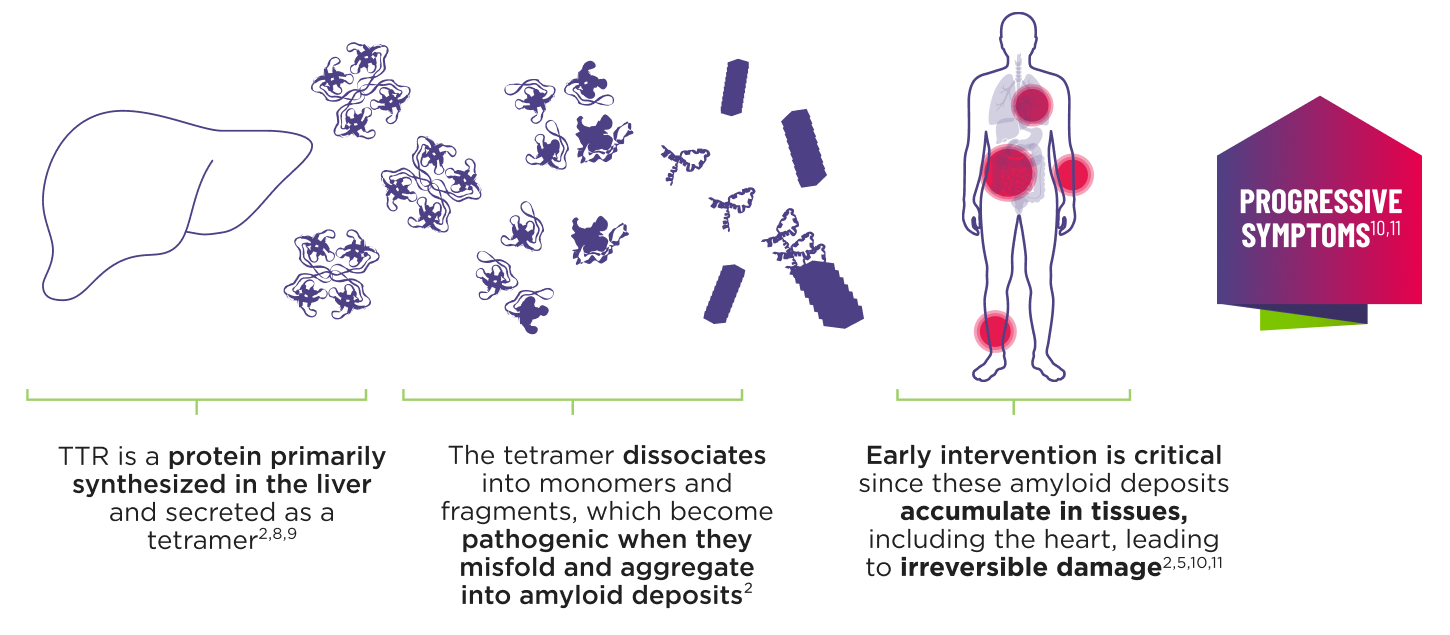
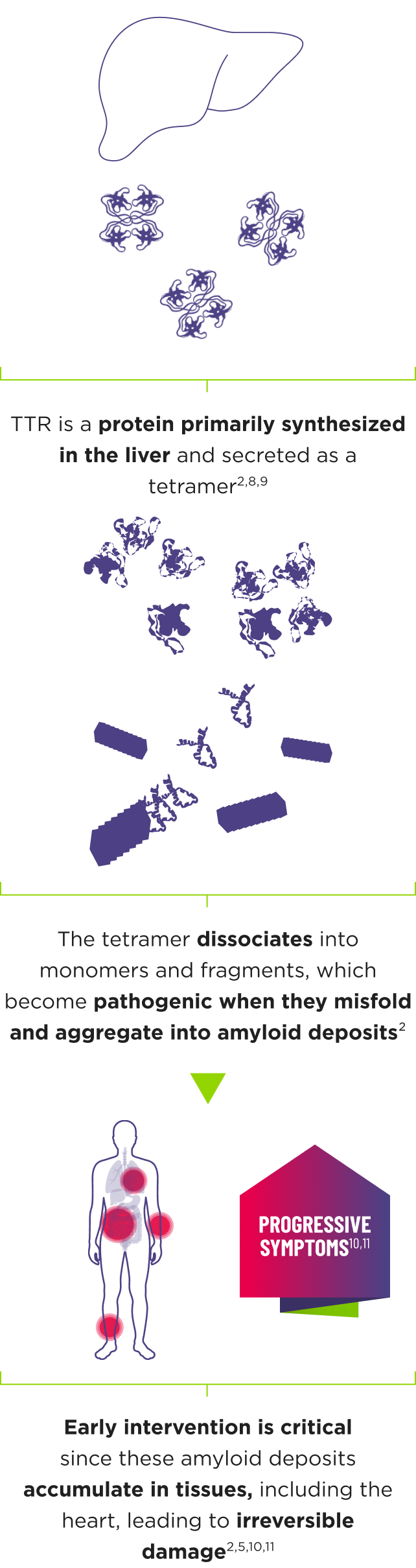
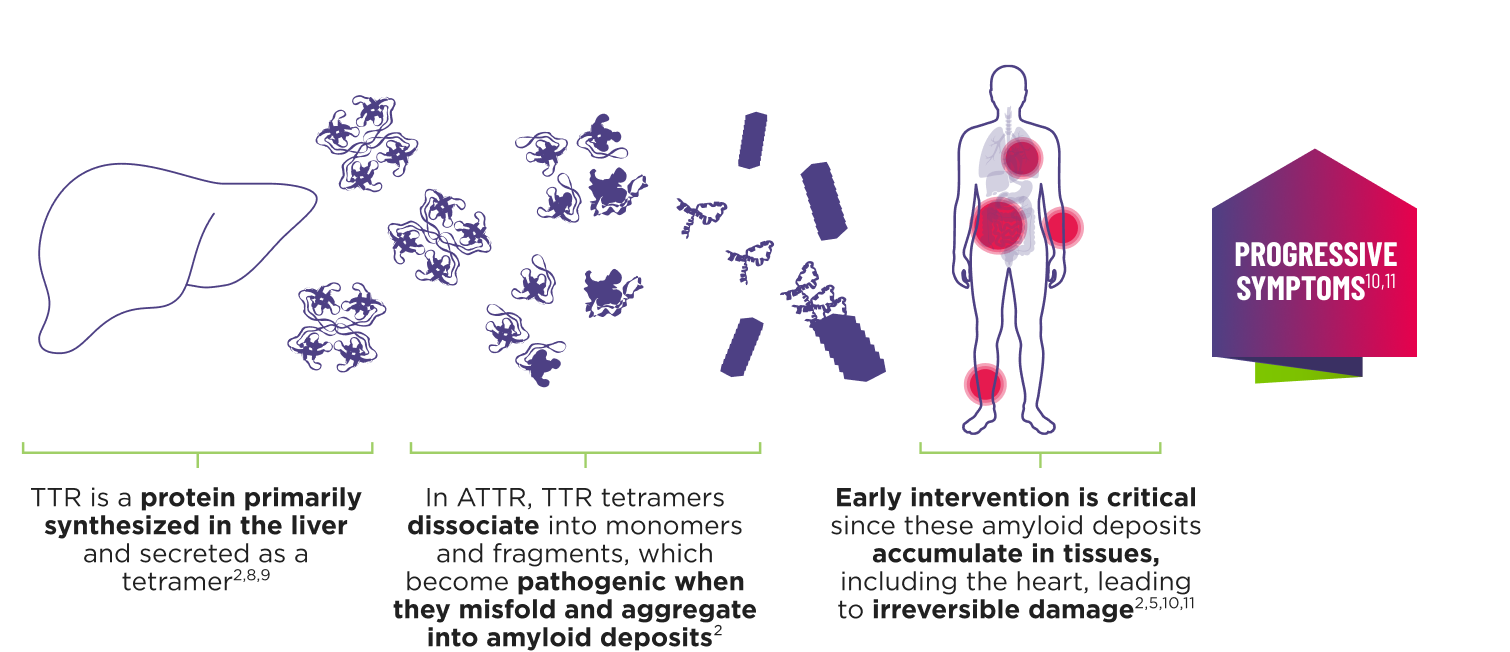
AMVUTTRA addresses the underlying cause of ATTR‑CM and hATTR‑PN with rapid knockdown of TTR1-3
Intervene early with AMVUTTRA, the first and only treatment approved for ATTR‑CM and hATTR‑PN1,21,22
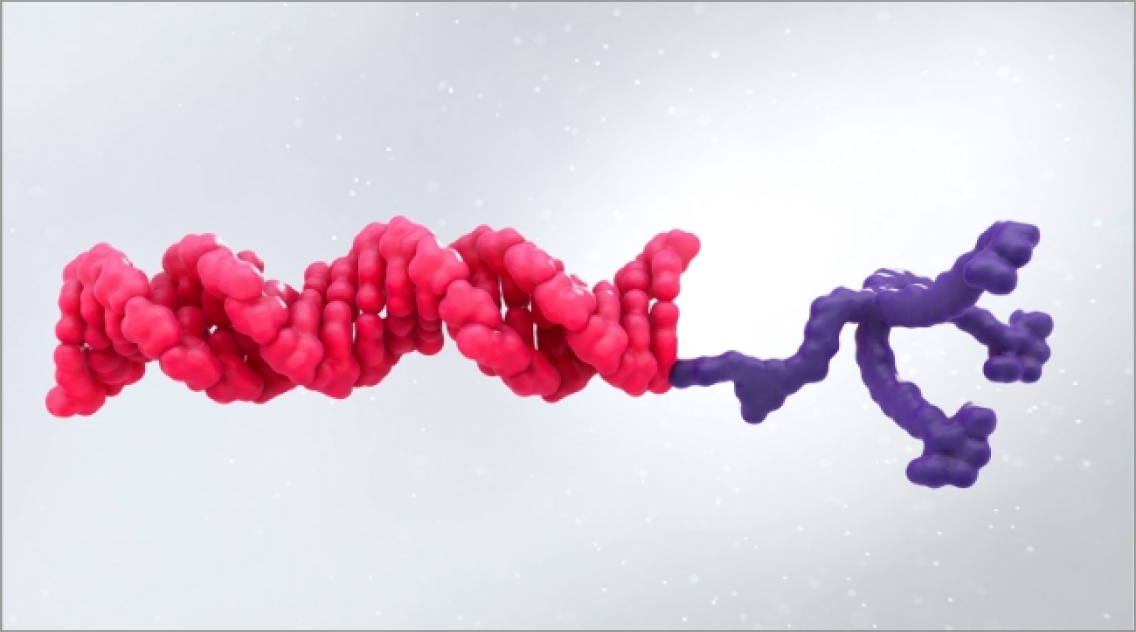
- AMVUTTRA is formulated for targeted delivery to the liver, the primary source of TTR production1,2
- AMVUTTRA deploys the body’s natural silencing complex to act upstream of tetramer formation1,20
ATTR-CM=cardiomyopathy of transthyretin-mediated amyloidosis; hATTR-PN=polyneuropathy of hereditary transthyretin-mediated amyloidosis; mRNA=messenger RNA; RNAi=ribonucleic acid interference; TTR=transthyretin.
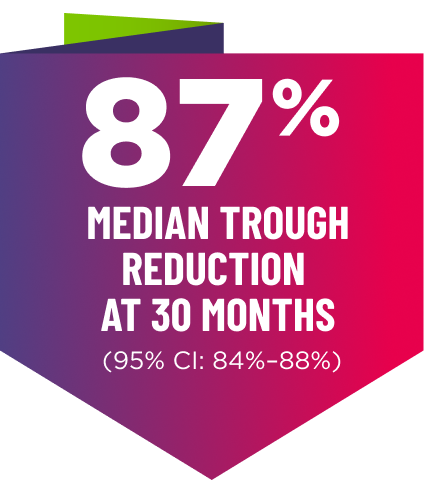
AMVUTTRA delivered RAPID KNOCKDOWN of TTR as early as 6 weeks in HELIOS-B3,23*
Knockdown of TTR was sustained through 30 months23†‡
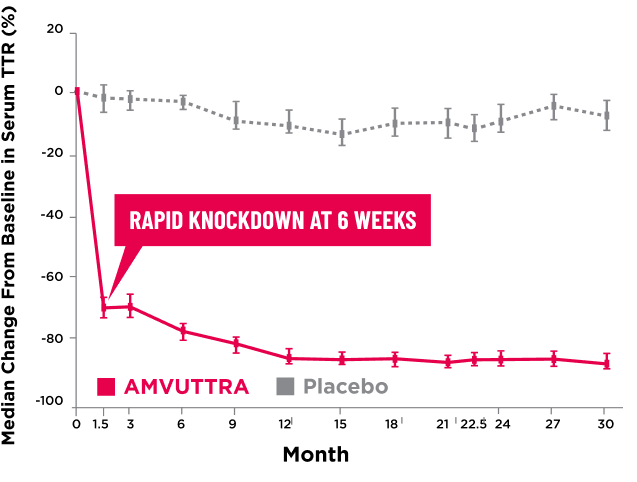
- In the HELIOS-B pivotal trial, serum TTR was evaluated in patients with ATTR-CM treated with 25 mg of AMVUTTRA via subcutaneous injection once every 3 months1
- A similar reduction in TTR levels was observed regardless of baseline tafamidis use, disease type (wtATTR or hATTR), sex, age, weight, or race1
TTR knockdown was first measured in the serum at 6 weeks in HELIOS-B. 23
TTR knockdown level is demonstrated through serum TTR reduction. 1
Bars indicate 95% confidence intervals. 23
CI=confidence interval; hATTR=hereditary ATTR; TTR=transthyretin; wtATTR=wild-type ATTR.
AMVUTTRA achieves sustained knockdown of TTR with a subcutaneous injection 4 times per year.1
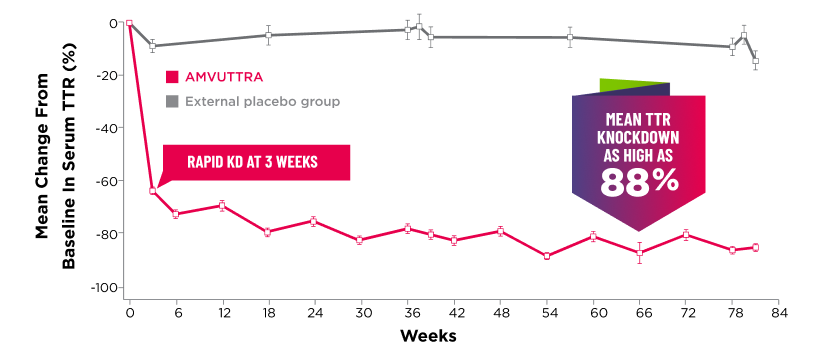
Treatment with AMVUTTRA led to rapid knockdown of TTR as early as 3 weeks in HELIOS-A24*
Knockdown of TTR was sustained over 18 months24,25†‡
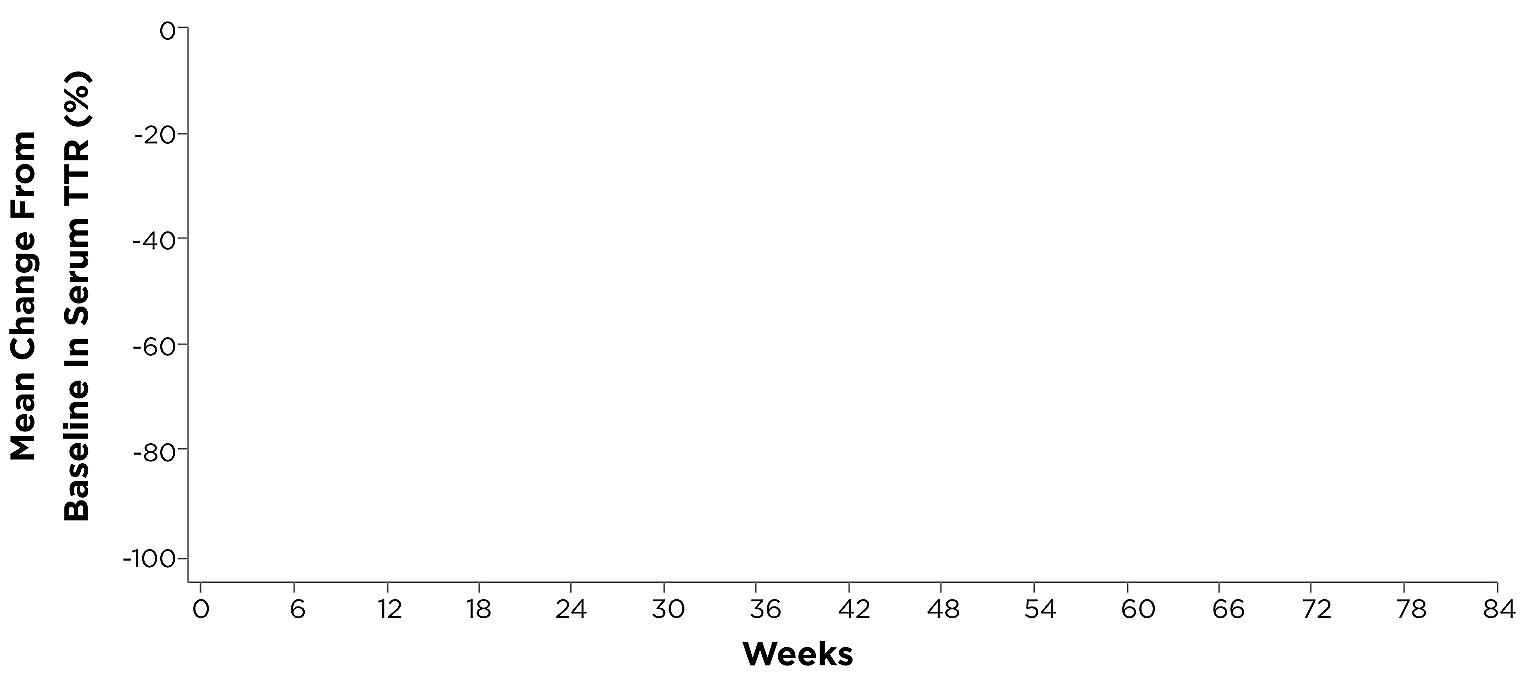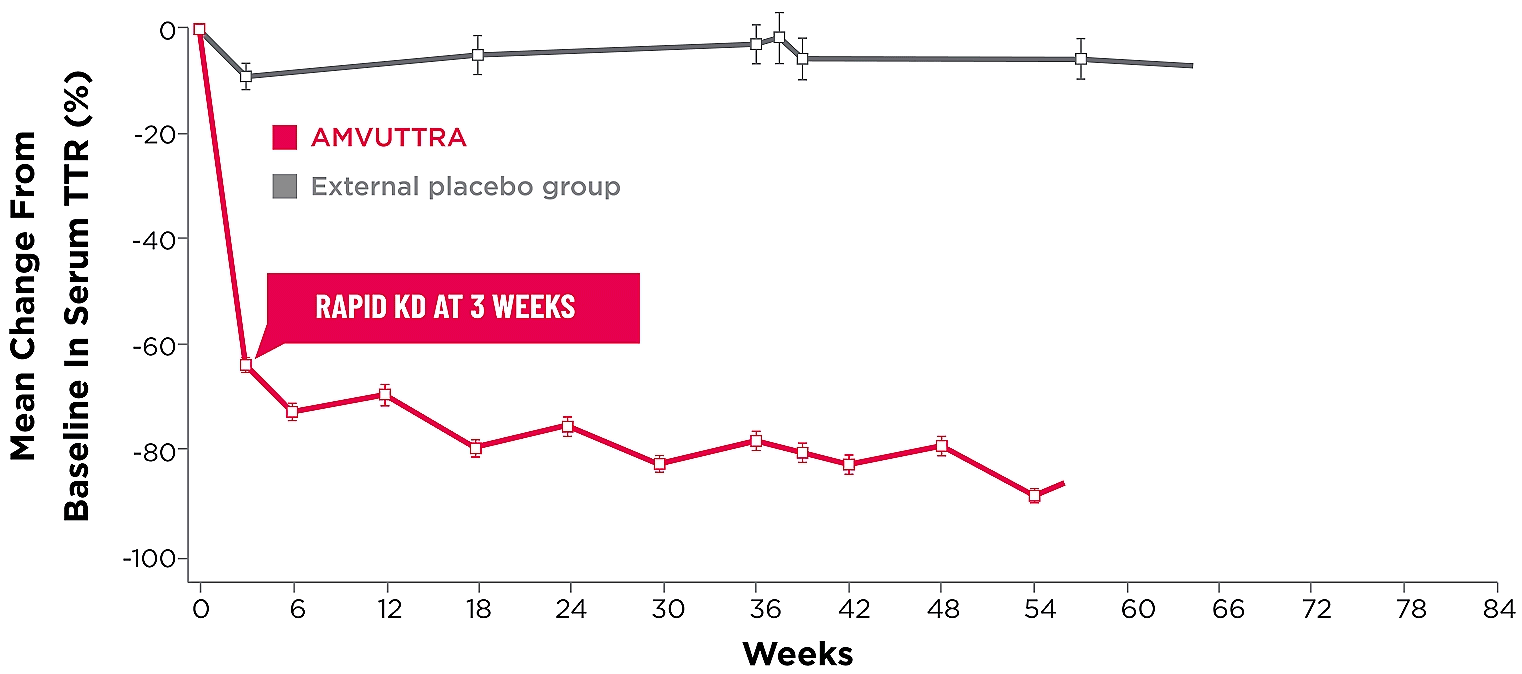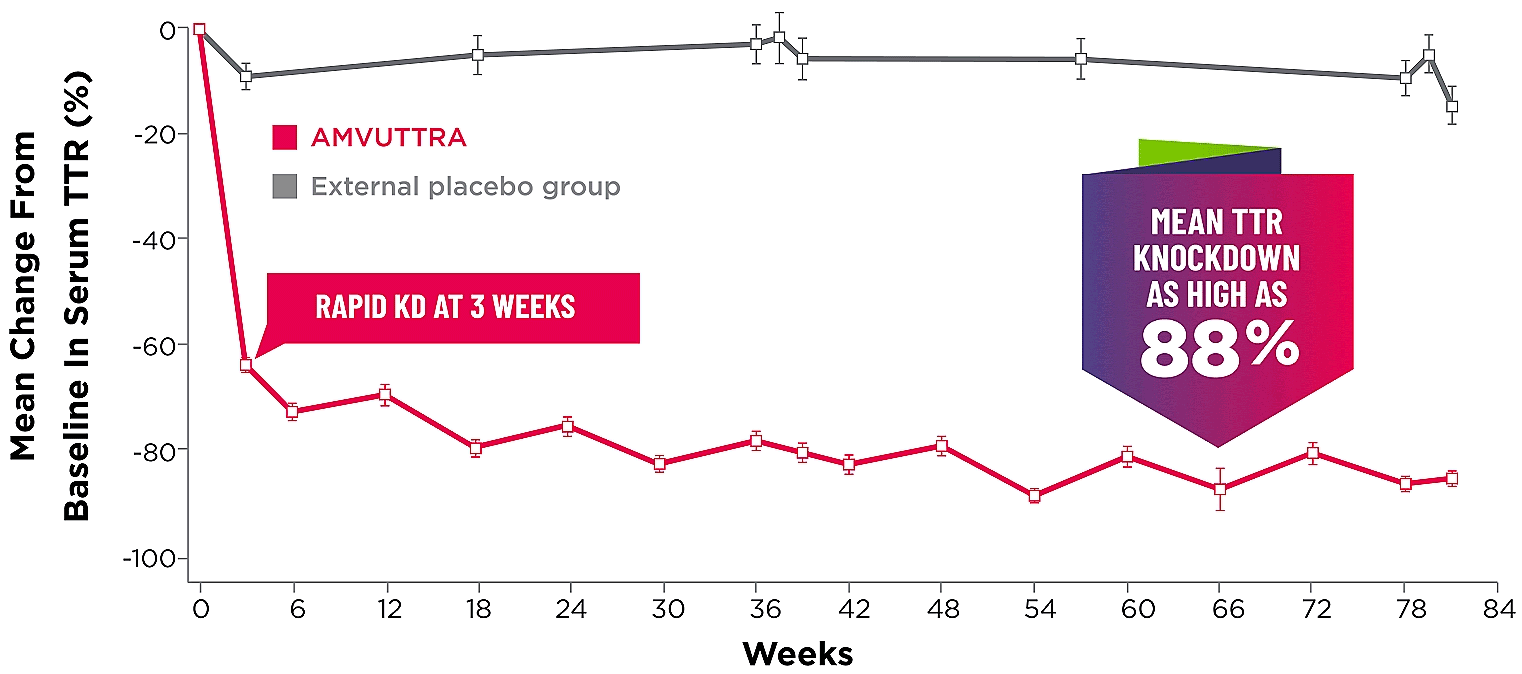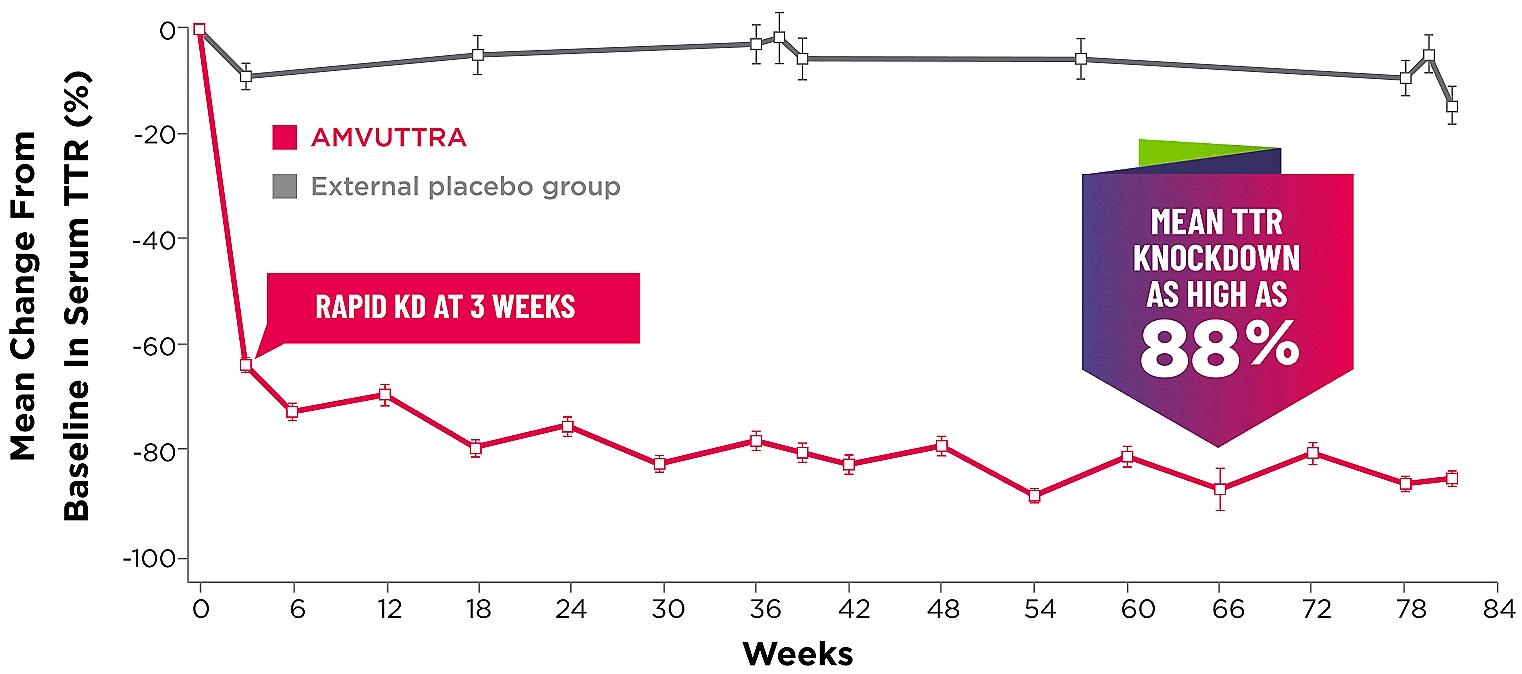
*First measured at 3 weeks.
†Bars represent SEM.
‡TTR knockdown level is demonstrated through serum TTR reduction.
KD=knockdown; SEM=standard error of the mean.
- Serum TTR was evaluated in patients with hATTR-PN treated with 25 mg AMVUTTRA via subcutaneous injection once every 3 months1
- With AMVUTTRA, similar rapid knockdown in serum TTR was observed regardless of V30M variant status, previous TTR stabilizer use, sex, age, weight, or race1,23