Safety
AMVUTTRA® has an established safety and tolerability profile in adults1
The safety and tolerability of AMVUTTRA were established in a study of adult patients with hATTR-PN (HELIOS-A).1
No new safety issues were identified in HELIOS-B. The frequency of pain in extremity, arthralgia, and dyspnea were similar to placebo.2
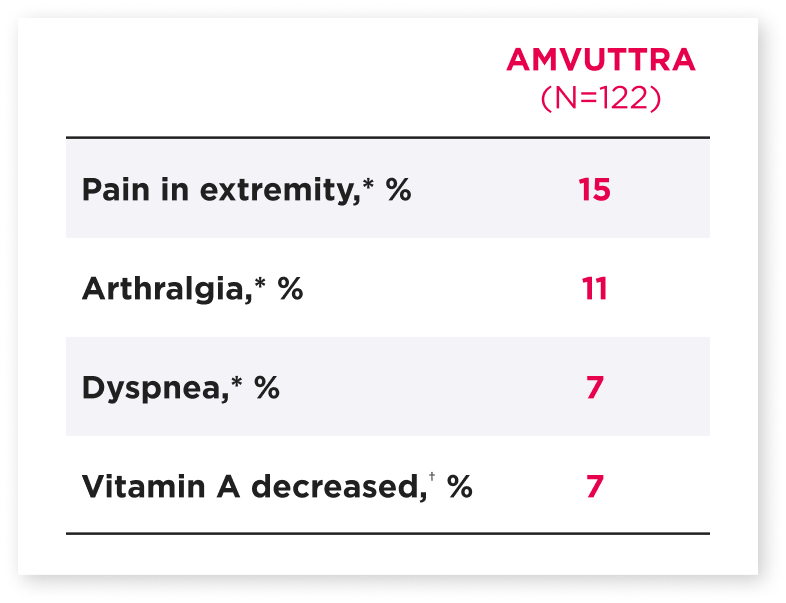
Safety in HELIOS-A (hATTR-PN)1

Safety in HELIOS-B (ATTR-CM)2


Comprised of several similar terms.1
Percentage only reflects those reported as an adverse reaction.1
No new safety concerns have been identified to date related to long-term reduction in TTR levels from Alnylam’s clinical studies, open-label extension studies, or post-marketing surveillance.2-6‡
Comprised of several similar terms.1
Percentage only reflects those reported as an adverse reaction.1
As of December 2025.

